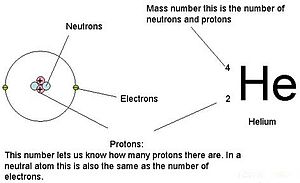
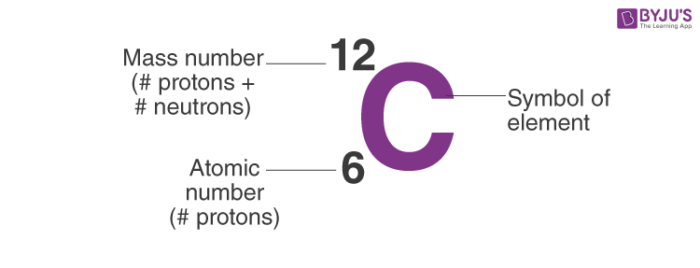
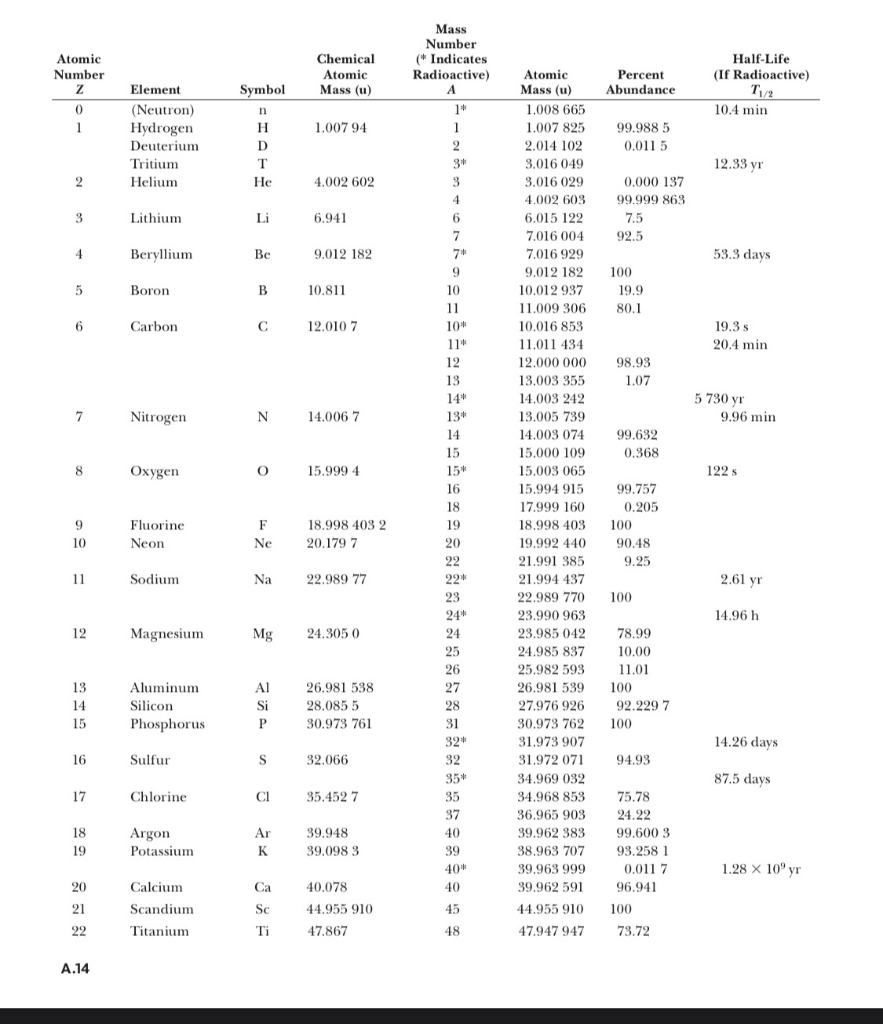
Atomic notation X A Z X = Symbol (C, Au) A = Atomic Mass Number = #nucleons (Protons + Neutrons) Z = Atomic Number = #protons C 12 6 Carbon A = 12, Z = - ppt download

Nuclear Chemistry Chapter 21 Slideshow and powerpoint viewer: Atomic number (Z) = number of protons in nucleus Mass number (A) = number of protons + number of neutrons = atomic number (Z) + number

Influence of the effective atomic number Z eff on the attenuation (HU).... | Download Scientific Diagram

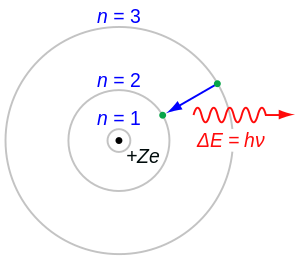
57. Consider an atom with atomic number Z as consisting of a positive point charge at the centre and surrounded by a distribution of negative electricity uniformly distributed within a sphere of


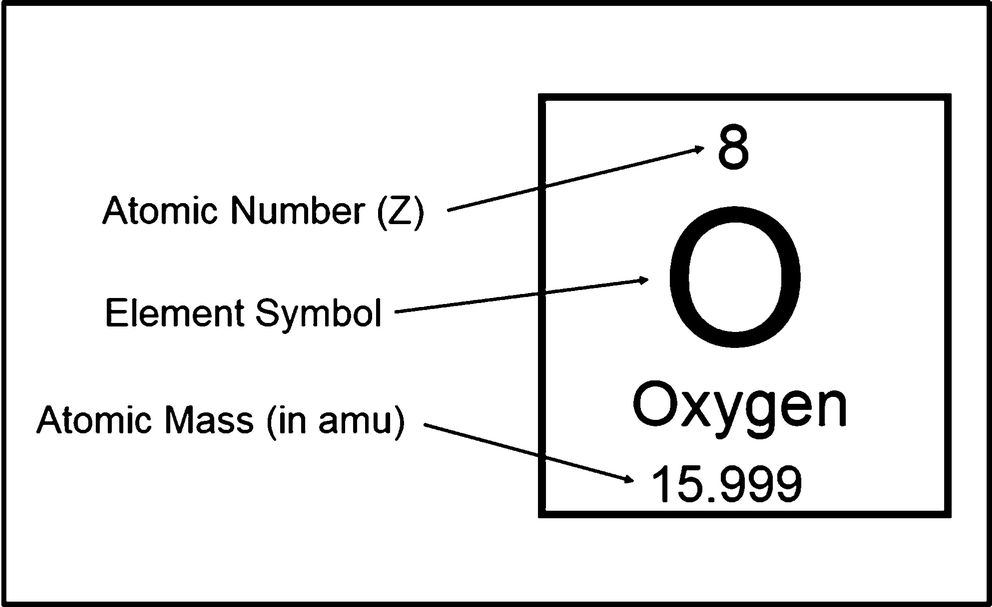
![SOLVED] Atomic number of a nucleus is Z and mass number is A. Find the nu - Self Study 365 SOLVED] Atomic number of a nucleus is Z and mass number is A. Find the nu - Self Study 365](https://storage.googleapis.com/tb-img/production/20/05/F1_J.K_8.05.2020_Shashi_D%203.png)